
SKU : G1221-5ML
หมวดหมู่ : Biochemicals , 1. Chemical and Reagents , Buffers , Servicebio ,
แบรนด์ : Servicebio
Share
Product Information
Product Name | Cat.No. | Spec. |
Tissue Autofluorescence Quencher | G1221-5ML | 2×5 mL |
Description
Immunofluorescence technology uses fluorescently labeled antibodies to locate antigens bound to them, realizing subcellular visual observation of proteins, glycan proteins, small biomolecules and non-biological molecules. During this process, autofluorescence in the tissue often seriously interferes with the observation. Autofluorescence is produced in the process of immunofluorescence test has nothing to do with the purpose of signal background fluorescence signal of a general designation, the reasons mainly from the composition of the organization itself, such as flavin, porphyrins, plant chlorophyll, collagen, elastin, red blood cells, fat brown pigment can produce strong spontaneous fluorescence, significantly influence the signal-to-noise ratio of immunofluorescence experiments. In addition, formalin, paraformaldehyde and other substances used in tissue fixation will also produce a lot of autofluorescence.
This product can effectively reduce tissue autofluorescence, improve the signal to noise ratio of immunofluorescence detection. The active component of liquid A was divalent copper ion, which could effectively reduce the autofluorescence of red blood cells. The active component of liquid B was Sudan black, which could significantly reduce the autofluorescence caused by lipofuscin.
Storage and Handling Conditions
Keep at room temperature and away from light, valid for 12 months.
Component
Component Number | Component | G1221-5ML | |
G1221-1 | Tissue Autofluorescence Quencher A | 5 mL | |
G1221-2 | Tissue Autofluorescence Quencher B | 5 mL | |
Product Manual | 1 pc | ||
Assay Protocol
1. Autofluorescence quenching before immunolabelling: After antigen repair, tissue sections were circled with histochemical strokes, then tissue autofluorescence quenching agent A was dropped to cover the tissue and incubated for 30 min at room temperature, and then washed with pure water for 5 min.
2. Routine immunofluorescence detection procedures including sealing, primary antibody incubation, secondary antibody incubation, and DAPI counterstaining were performed on tissue sections.
3. Autofluorescence quenching after immunolabelling: Tissue autofluorescence quenching agent B was dropped to cover the tissue and incubated for 5 min at room temperature in darkness, then rinsed with running water for 3 min.
4. Sealing: The glass slide is placed in PBS and washed by shaking (it can be placed on the decolorization shaker) for 3 times, 5 min each time. The slices were slightly dried and sealed for microscopic examination.
Note:
1. The principle of autofluorescence in different samples is different, and the effect of tissue autofluorescence quencher may be different.
2. Theoretically, quenchers for autofluorescence will also reduce the fluorescence intensity of antibodies to a certain extent. It was found that the quenching degree of autofluorescence by this reagent was far more than the decrease of antibody fluorescence intensity, and there was a good balance between the two.
3.The tissue autofluorescence quenching reagent A is used in conjunction with hydrogen peroxide, which
can affect DAPI staining. If performing TSA experiments, it is not recommended to add the tissue autofluorescence quenching reagent A.
4. After adding tissue autofluorescence quencher B liquid, the tissue will be stained dark blue, but it will not affect the fluorescence photography.
5. After incubation with liquid B, the sections can be washed with water or PBS, but not with the solution containing Tween, otherwise, the excessive washing of liquid B will affect the background fluorescence quenching effect.
6. Tighten the cap of tissue autofluorescence quencher B after each use to prevent solvent volatilization.
7.If there are impurities on the tissue after incubation with solution B, it can be diluted by half with 70%
ethanol before use.
8. Wear a lab coat and disposable gloves during operation.
Image:
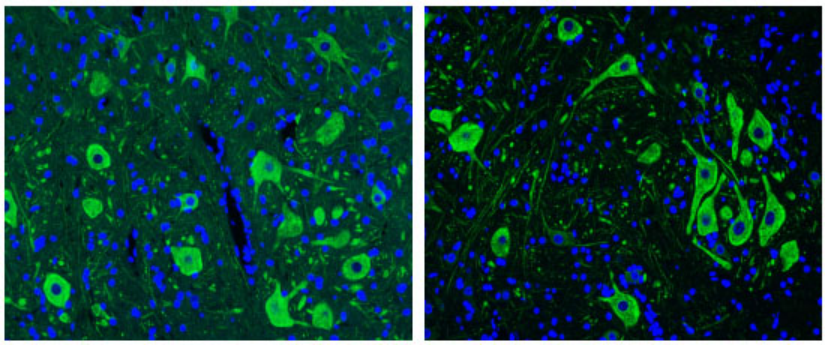
Left: Paraffin sections of spinal cord tissue for NF200; Right: NF200 was made after the same slice was treated with this reagent.
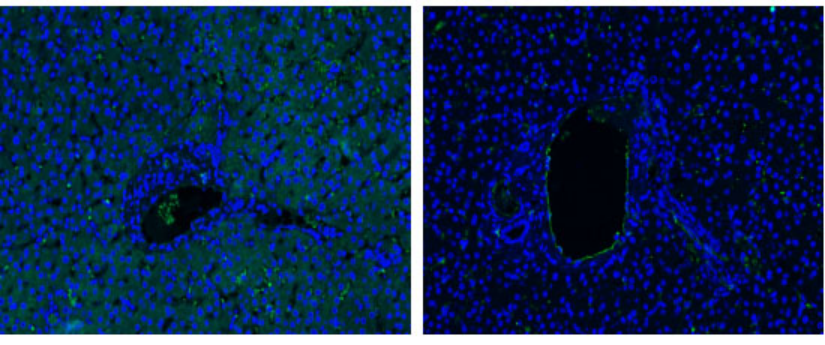
Left: paraffin sections of liver tissue for CD31; Right: The same slice was treated with this reagent before CD31.
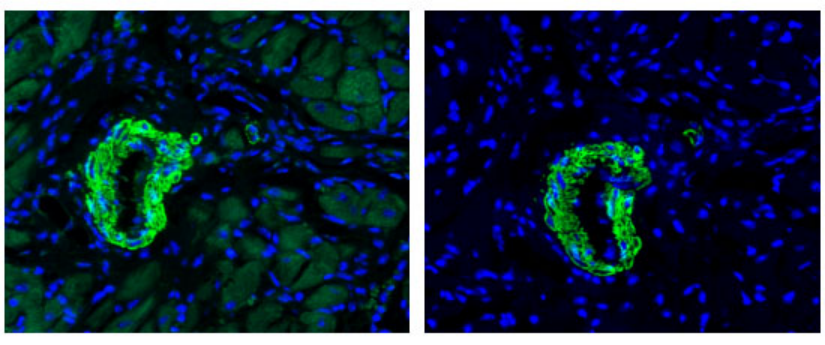
Left: Paraffin sections of rat myocardial tissue for α-SMA; Right: The same slice was processed with this reagent before α-SMA.
For Research Use Only!